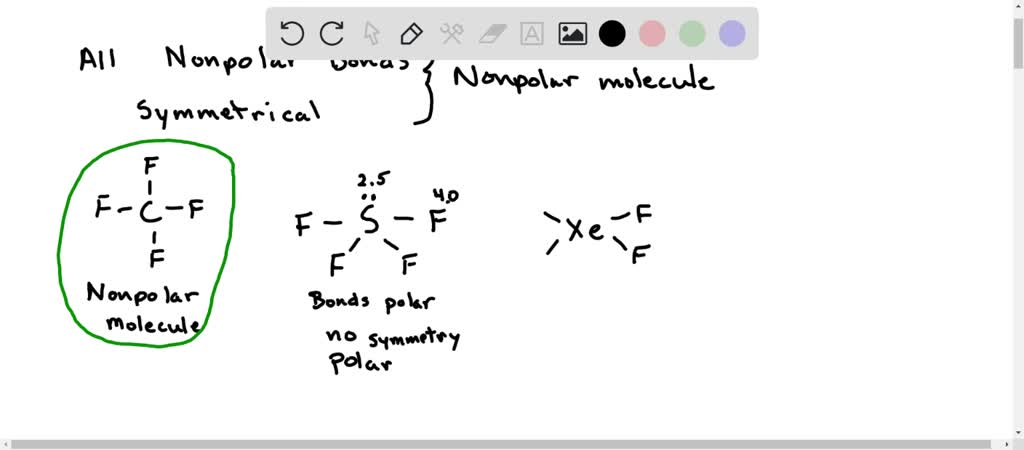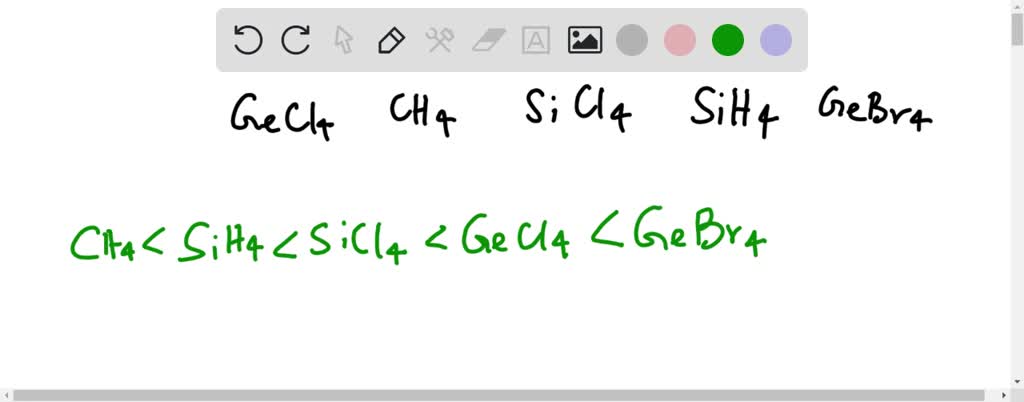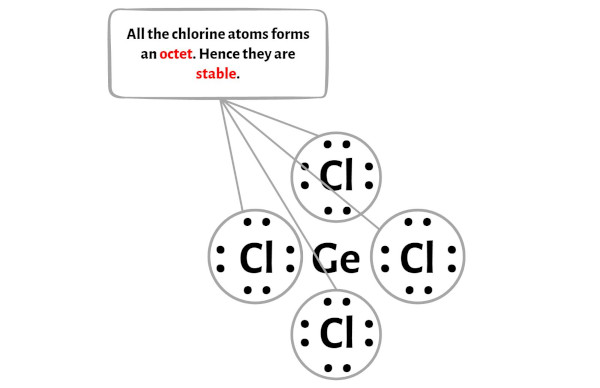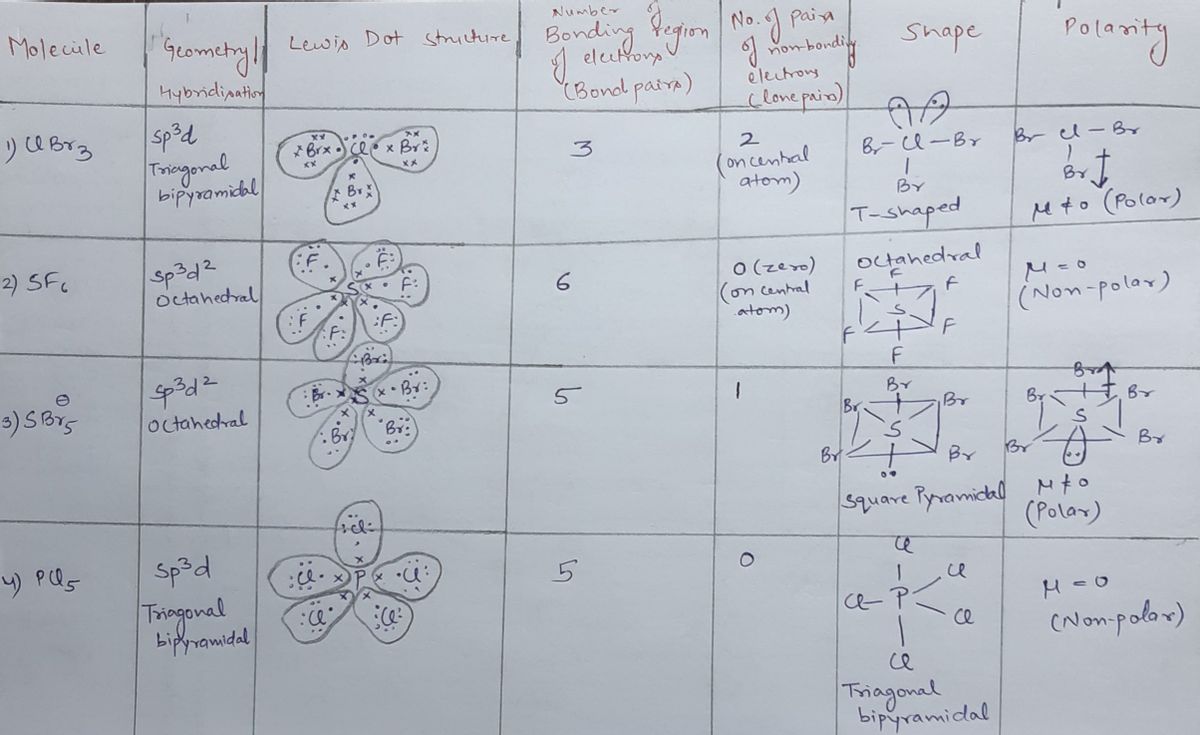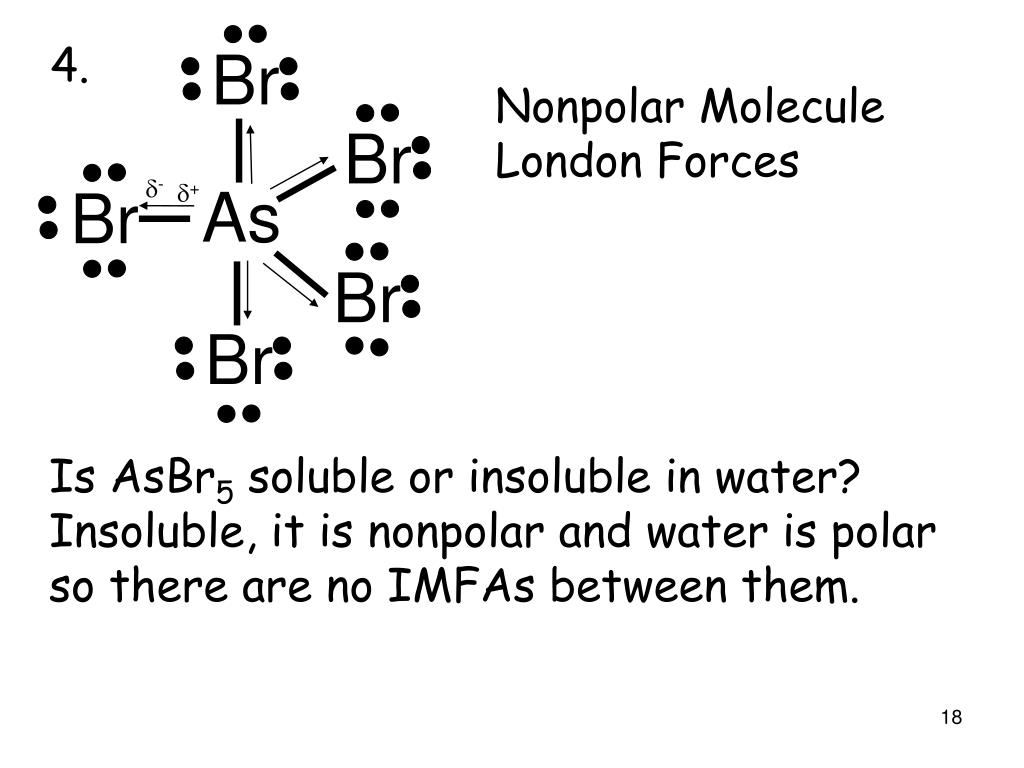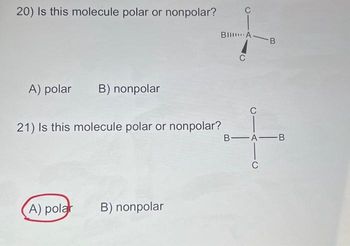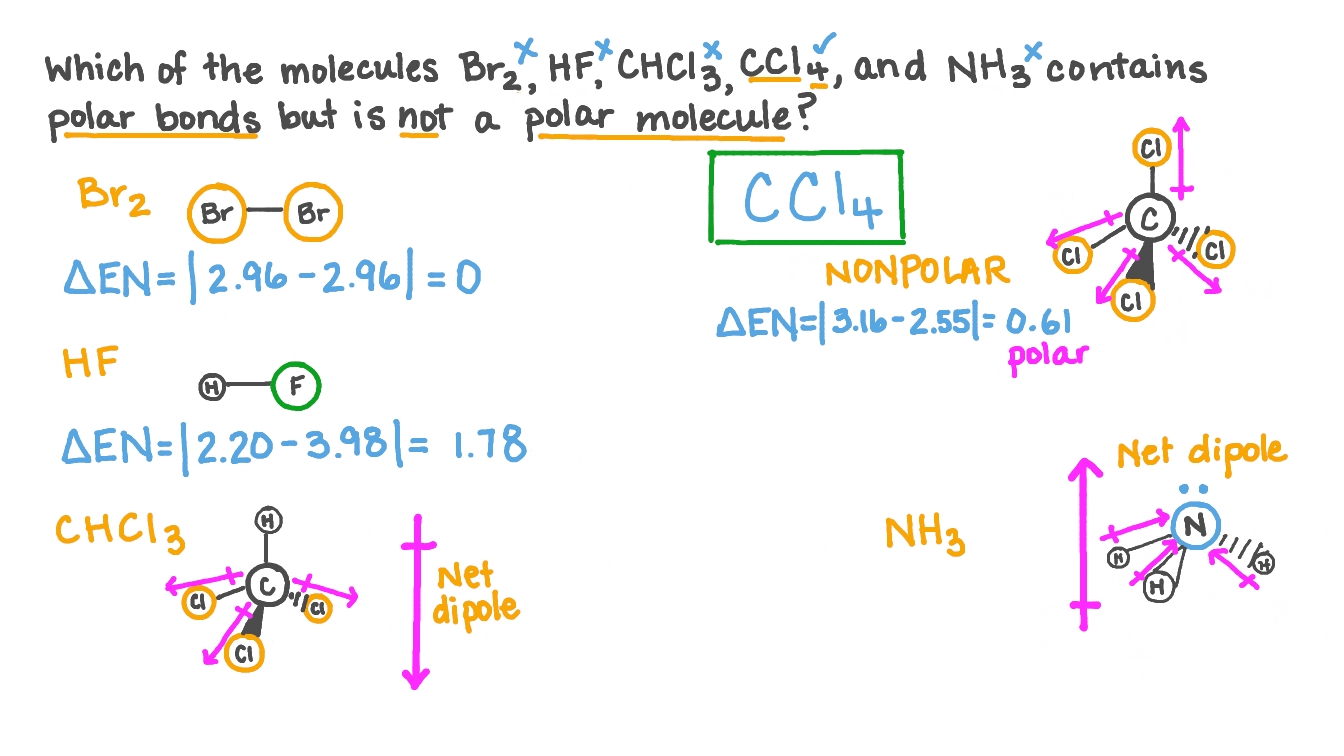
Question Video: Determining the Molecule That Contains Polar Bonds but Is Not a Polar Molecule | Nagwa

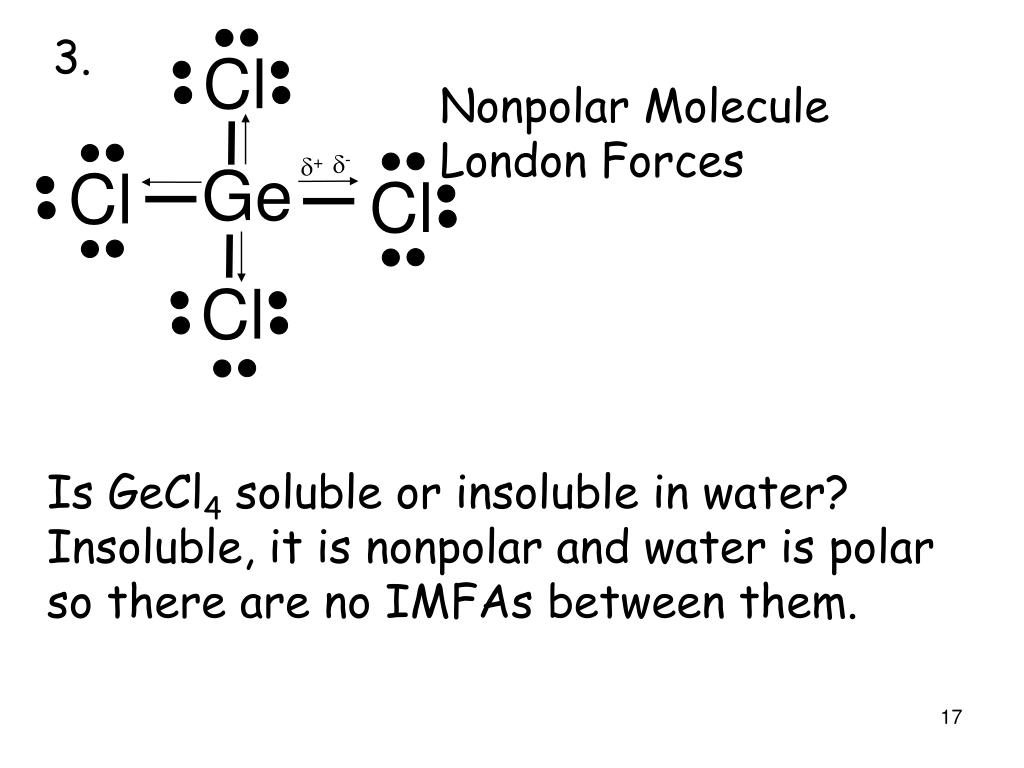
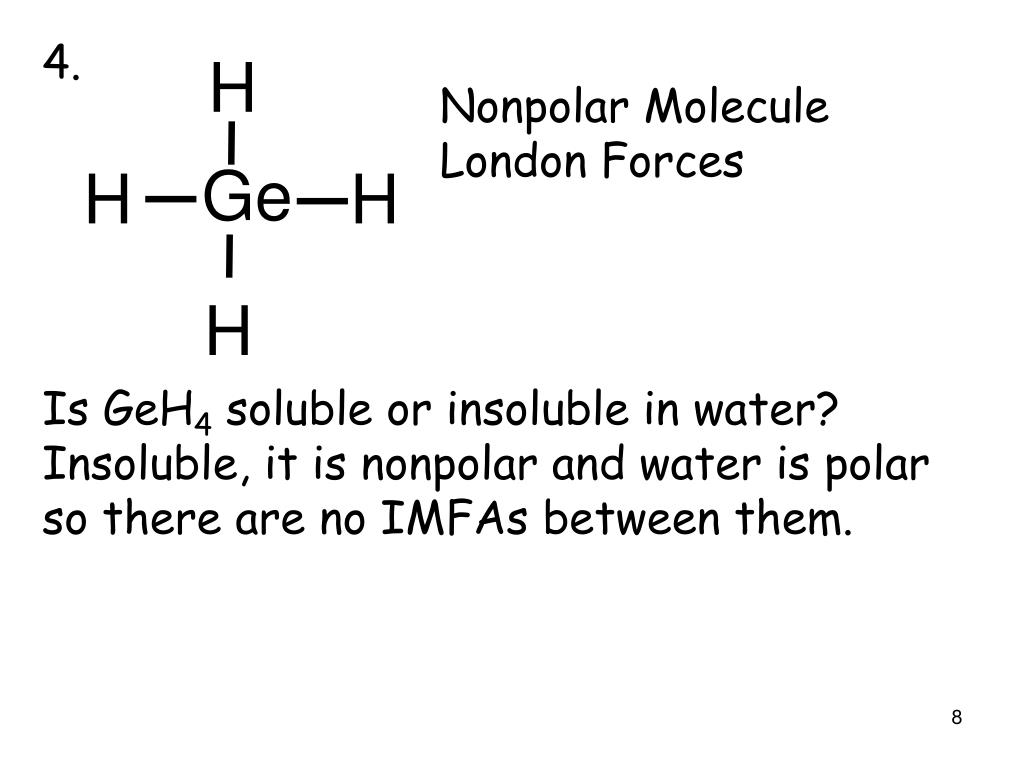
SOLVED: Polar Nonpolar HCl Most Stable Electron Pair Molecular Geometry Perspective Sketch State Bond Angles Lewis Dot Structure Other Resonance Structures K2SO4 ICl3 GeCl4 SCl2 TeO2 O2

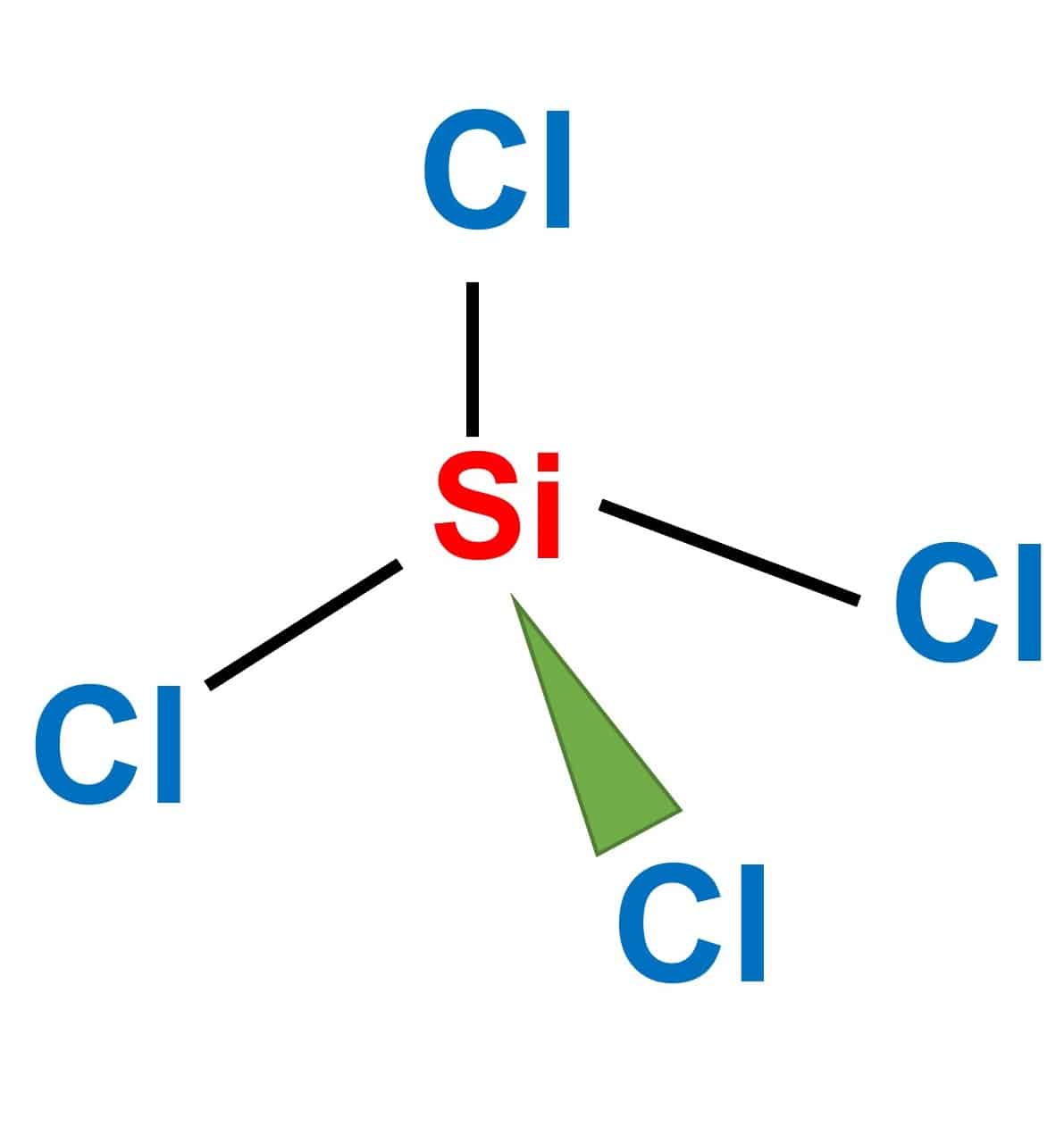
Is SiCl4 Polar or Non-polar? (Silicon Tetrachloride) | Is SiCl4 Polar or Non-polar? (Silicon Tetrachloride) We know the concept of polarity is a bit tricky and one might need a little practice

SOLVED: What formula do you predict for the ionic compound that forms between Ge and N? (3 pts) GeN GezN Ge-Na Ge;Nz Germanium dioxide; GeO2, is a hard solid that is soluble